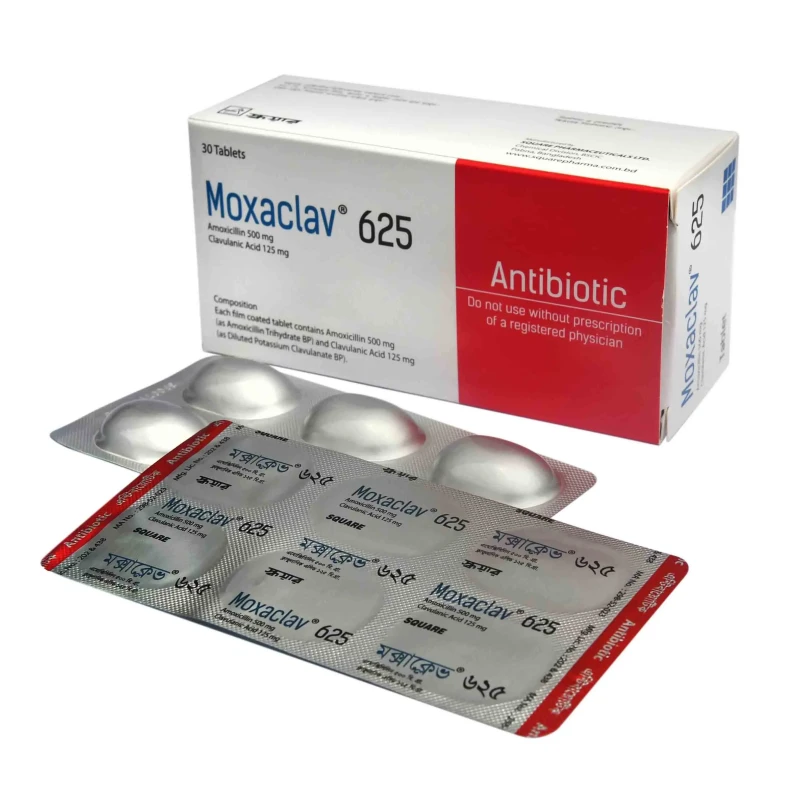

Moxaclav Tablet, Amoxicillin + Clavulanic Acid 500 mg+125 mg
Inhouse product
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00
Reviews & Ratings
Indications
Co-amoxiclav is
indicated for short-term treatment of bacterial infections at the following
sites:
- Upper respiratory tract
infections (including ENT) e.g.tonsillitis,sinusitis,otitis media.
- Lower respiratory tract infections
e.g.acute and chronic bronchitis, lobar and bronchopneumonia.
- Genito-urinary tract infections
e.g.cystitis,urethritis,pyelonephritis.
- Skin and soft tissue
infections.
- Bone and joint infections
e.g.osteomyelitis.
- Other infections e.g.septic
abortion,puerperal sepsis,intra-abdominal sepsis etc.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Composition
- 375 mg tablet: Each film coated tablet contains Amoxicillin 250 mg
as Amoxicillin Trihydrate BP and Clavulanic Acid 125 mg as Diluted
Potassium Clavulanate BP.
- 625 mg tablet: Each film coated tablet contains Amoxicillin 500 mg
as Amoxicillin Trihydrate BP and Clavulanic Acid 125 mg as Diluted
Potassium Clavulanate BP.
- 1 gm tablet: Each film coated tablet contains Amoxicillin 875 mg
as Amoxicillin Trihydrate BP and Clavulanic Acid 125 mg Diluted Potassium
Clavulanate BP.
- Powder for
Suspension: Each 5 ml reconstituted
suspension contains Amoxicillin 125 mg as Amoxicillin Trihydrate BP and
Clavulanic Acid 31.25 mg as Diluted Potassium Clavulanate BP.
- Powder for
Suspension (Forte): Each 5
ml reconstituted suspension contains Amoxicillin 400 mg as Amoxicillin
Trihydrate BP and Clavulanic Acid 57.5 mg as Diluted Potassium Clavulanate
BP.
- 1.2 gm Injection: Each vial contains sterile mixture of Amoxicillin
Sodium BP (equivalent to Amoxicillin 1 gm) and Clavulanate Potassium USP
(equivalent to Clavulanic Acid 200 mg).
- 0.6 gm injection: Each vial contains sterile mixture of Amoxicillin
Sodium BP (equivalent to Amoxicillin 500) and Clavulanate Potassium USP
(equivalent to Clavulanic Acid 100 mg).
Pharmacology
Pharmacodynamic
properties: Co-amoxiclav is an
antibacterial combination consisting of the antibiotic Amoxicillin and the
(3-lactamase inhibitor Clavulanic Acid. Amoxicillin has a broad spectrum of
bactericidal activity against many Gram-positive & Gram-negative
microorganisms but it is susceptible to degradation by (3-lactamases and
therefore the spectrum of activity does not include microorganisms, which
produce these enzymes. Clavulanic acid possesses the ability to inactivate a
wide range of beta-lactamase enzymes commonly found in microorganisms resistant
to penicillins and cephalosporins. Thus Clavulanic acid in this preparation
protects Amoxicillin from degradation by (3-lactamase enzymes and effectively
extends the antibiotic spectrum to embrace a wide range of microorganisms.
Pharmacokinetic
properties: The pharmacokinetics
of the two components of Co-amoxiclav is closely matched. Peak serum levels of
both occur about one hour after oral administration. Absorption of Co-amoxiclav
is optimized at the start of a meal. Both clavulanate and Amoxicillin have low
levels of serum binding; about 70% remains free in the serum. Doubling the
dosage of Co-amoxiclav approximately doubles the serum levels achieved.
Dosage
Adults and children
over 12 years:
Tablet:
- The usual adult dose is one 625
mg Tablet every 12 hours or one 375 mg Tablet every 8 hours.
- For more severe infections and
infections of the respiratory tract, the dose should be one 1 gm Tablet
every 12 hours or one 625 mg Tablet every 8 hours.
Suspension:
- Children 6-12 years: 2
teaspoonful every 8 hours.
- Children 1-6years: 1
teaspoonful every 8 hours.
- Children below 1 year: 25
mg/kg/day in divided doses every 8 hours, for example a 7.5 kg child would
require 2 ml suspension t.i.d, Treatment should not be extended beyond 14
days without review.
Forte
suspension:
- The usual recommended daily
dosage: 25/3.6 mg/kg/day in mild to moderate infections (upper respiratory
tract infections e.g. recurrent tonsilitis, lower respiratory infections,
and skin and soft tissue infections)।
- For serious infections: 45/6.4
mg/kg/day for the treatment of more serious infections (upper respiratory
tract infections, e.g. otitis media and sinusitis, lower respiratory
infections e.g. bronchopneumonia, and urinary tract infections).
Children of 2 to 12
years:
Mild to moderate infections:
- 25/3.6 mg/kg/day (Suspension)
- 2-6 years (13-21 kg) 2.5 ml
suspension b.i.d
- 7-12years (22-40kg) 5 ml
suspension b.i.d
Serious infections:
- 45/6.4 mg/kg/day (Forte
Suspension)
- 2-6 years (13-21 kg) 5 ml
suspension b.i.d
- 7-12 years (22-40 kg) 10 ml
suspension b.i.d
IV
Injection
Adults-
- Usually, 1.2 gm every 8 hours
- Increased in more serious
infections to 1.2 gm every 6 hours
- For surgical prophylaxis: The
usual dose is 1.2 gm at induction, for high risk procedures (eg. colorectal
surgery) up to 2-3 gm may be given every 8 hours.
Children-
- 0 to 3 months: 30 mg/kg every 8
hours. (every 12 hours in the perinatal period and in premature infants.
- 3 months to 12 years: Usually
30 mg/kg every 8 hours increased in more serious infection to 30 mg/kg
every 6 hours.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Administration
Oral dosage form: This
may be taken without regard to meals; however, absorption of clavulanate
potassium is enhanced when Amoxicillin and Clavulanic acid are administered at
the start of a meal. To minimize the potential for gastrointestinal intolerance,
Amoxicillin and Clavulanic acid should be taken at the start of the meal.
IV injection is not suitable for intramuscular or subcutaneous administration.
The reconstituted vial can be administered intravenously by injection (over 2
minutes) or slow intravenous infusion (30 minutes). The contents of the content
of the vial must be used within 20 minutes and thereafter any unused material
should be discarded.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Interaction
Prolongation of
bleeding time and prothrombin time have been reported in some patients
receiving Co-amoxiclav. In common with other broad-spectrum antibiotics,
Co-amoxiclav may reduce the efficacy of oral contraceptives and patients should
be warned accordingly. Concomitant use of allopurinol during treatment with
amoxicillin can increase the likelihood of allergic skin reactions. There are
no data on the concomitant use of Co-amoxiclav and allopurinol.
Contraindications
History of Penicillin
hypersensitivity. Attention should be paid to possible cross-sensitivity with
other beta-lactam antibiotics e.g. cephalosporins. Also contraindicated for
patients with a previous history of Co-amoxiclav or Penicillin-associated
cholestatic jaundice.
Side Effects
Side effects, as with
Amoxicillin, are uncommon and mainly of a mild and transitory nature.
Diarrhoea, pseudomembranous colitis, indigestion, nausea, vomiting and
candidiasis have been reported, if gastrointestinal side effects occur with
oral therapy, that may be reduced by taking Co-amoxiclav at the start of meals.
Hepatitis and cholestatic jaundice have been reported rarely but are usually
reversible. Urticarial and erythematous rashes sometimes occur. Rarely erythema
multiforme, Stevens-Johnson Syndrome and exfoliative dermatitis have been
reported. In common with other beta-lactam antibiotics, angioedema and
anaphylaxis have been reported.
Pregnancy & Lactation
Animal studies with
orally and parenterally administered Co-amoxiclav have shown no teratogenic
effect. The drug has been used orally in human pregnancy in a limited number of
cases with no untoward effect; however, the use of Co-amoxiclav in pregnancy is
not recommended unless considered essential by the physician. During lactation,
trace quantities of Amoxicillin can be detected in breast milk.
Precautions & Warnings
Co-amoxiclav should be
used with care in patients on anticoagulation therapy or with severe hepatic
dysfunction. In patients with moderate or severe renal impairment, dosage
should be adjusted. During the administration of a high dose of Co-amoxiclav
adequate fluid intake and urinary output should be maintained to minimize the
possibility of crystalluria.
Use in Special Populations
The dose should be
adjusted in case of patients with renal impairment
Adult:
- Mild impairment (Creatinine
clearance> 30ml/minute): No changein dosage.
- Moderate impairment (Creatinine
clearance 10-30 ml/minute): One 375 Tablet or one 625 Tablet 12 hourly or
1.2 gm IV followed by 0.6 gm IV 12 hourly.
- Severe impairment (Creatinine
clearance <10 ml/minute): Not more than one 375 mg tablet 12 hourly or
1.2 gm IV followed by 0.6 gm IV 24 hourly. Dialysis decreases serum
concentrations of Moxaclav and an additional 0.6 gm IV dose may need to be
given during dialysis and at the end of dialysis.
Children:
- A similar reduction in dosage
should be made for children.
- Administration hepatic
impairment: Dose with caution; monitor hepatic function at regular
intervals.
Overdose Effects
Problems of overdose
with Co-amoxiclav are unlikely to occur, if encountered gastrointestinal
symptoms and disturbance of the fluid and electrolyte balances may be evident.
Co-amoxiclav may be removed from the circulation by haemodialysis.
Therapeutic Class
Broad spectrum
penicillins
Reconstitution
IV injection: 1.2 gm IV injection can be reconstituted by
dissolving the powder in 20 ml Water for Injection BP. This IV injection should
not be reconstituted or mixed with: Dextrose solution, Sodium Bicarbonate
solution for injection, Protein Hydrolysates or other Proteinaceous fluids,
blood or plasma, Intravenous lipids. However, the reconstituted solution may be
injected into the drip tubing of infusion fluids containing glucose,
bicarbonate and dextran over a period of 3-4 minutes.
Storage Conditions
This should be stored
below 25°C, protected from light and moisture. Once reconstituted suspension
should be kept in the refrigerator (but not frozen) and should be usedby 7
days. Once reconstituted vial must be used within 20 minutes.
Frequently Bought Products
Flugal Capsule, Fluconazole 50 mg
Nulanso Capsule (Enteric Coated), Dexlansoprazole 60 mg
Genacaft Ophthalmic Solution 5 ml drop, Alcaftadine 0.25%
Ace IV Infusion 100ml, Paracetamol 10 mg/ml
Tridyl Tablet, Trihexyphenidyl Hydrochloride 5 mg
Entafi Capsule, Finasteride + Tadalafil 5 mg+5 mg
Neumig Tablet, Mirogabalin Besylate 2.5 mg
Product Queries (0)
Login Or Registerto submit your questions to seller
Other Questions
No none asked to seller yet
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00