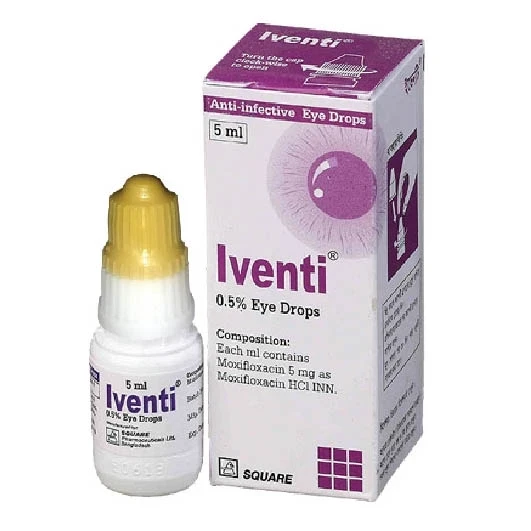

Iventi Ophthalmic Solution 5ml bottle, Moxifloxacin Hydrochloride 0.5%
Inhouse product
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00
Reviews & Ratings
Indications
Iventi ophthalmic
solution is indicated for the treatment of bacterial conjunctivitis caused by
susceptible strains of the following organisms:
- Aerobic Gram-positive microorganisms: Corynebacterium species, Micrococcus luteus,
Staphylococcus aureus, Staphylococcus epidermidis, Staphylococcus
haemolyticus, Staphylococcus hominis, Staphylococcus warneri,
Streptococcus pneumoniae, Streptococcus viridans group.
- Aerobic Gram-negative microorganisms: Acinetobacterlwoffii, Haemophilusinfluenzae,
Haemophilus parainfluenzae.
- Other microorganisms: Chlamydia trachomatis.
Intraocular injection: A highly effective Intraocular Antibiotic to
prevent Postoperative Bacterial Endophthalmitis.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Pharmacology
The antimicrobial action of Moxifloxacin
results from inhibition of the topoisomerase II (DNA gyrase) and topoisomerase
IV. DNA gyrase is an essential enzyme that is involved in the replication,
transcription and repair of bacterial DNA. Topoisomerase IV is an enzyme known
to play a key role in the partitioning of the chromosomal DNA during bacterial
cell division.
Dosage & Administration
Eye Drops: One drop in the affected eye 3 times per day for 7 days.
Eye Ointment: It should be applied thinly and evenly to
the affected eye three times a day for the first two days and for the next five
days apply two times a day or as advised by the registered physician.
Intraocular injection: Intraocular Moxifloxacin can be administered
as 0.2-0.4 ml dose prior to ocular surgery via intraocular route. The final
dose must be determined by the doctor.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Interaction
Drug-drug interaction studies have not been
conducted with Iventi ophthalmic solution. In vitro studies indicate that
Iventi does not inhibit CYP3A4, CYP2D6, CYP2C9, CYP2C19, or CYP1A2 indicating
that Iventi is unlikely to alter the pharmacokinetics of drugs metabolized by
these cytochrome P450 isozymes.
Contraindications
Moxifloxacin Hydrochloride ophthalmic solution
is contraindicated in patients with a history of hypersensitivity to
Moxifloxacin, to other quinolones, or to any of the components in this
medication.
Side Effects
The most frequently reported ocular adverse
events were conjunctivitis, decreased visual acuity, dry eye, keratitis, ocular
discomfort, ocular hyperemia, ocular pain, ocular pruritus, subconjunctival
hemorrhage, and tearing. These events occurred in approximately 1-6% of
patients. Nonocular adverse events reported at a rate of 1-4% were fever,
increased cough, infection, otitis media, pharyngitis, rash, and rhinitis.
Pregnancy & Lactation
Since there are no adequate and
well-controlled studies in pregnant women, Moxifloxacin Hydrochloride
ophthalmic solution should be used during pregnancy only if the potential
benefit justifies the potential risk to the fetus. Moxifloxacin has not been measured
in human milk, although it can be presumed to be excreted in human milk.
Caution should be exercised when Moxifloxacin hydrochloride ophthalmic solution
is administered to a nursing mother.
Precautions & Warnings
As with other anti-infectives, prolonged use
may result in overgrowth of nonsusceptible organisms, including fungi. If
superinfection occurs, discontinue use and institute alternative therapy.
Whenever clinical judgment dictates, the patient should be examined with the
aid of magnification, such as slit lamp biomicroscopy, and, where appropriate,
fluorescein staining. Patients should be advised not to wear contact lenses if
they have signs and symptoms of bacterial conjunctivitis.
Therapeutic Class
Ophthalmic antibacterial drugs
Storage Conditions
Store bellow 25°C. Do not freeze. Store in
cool and dry place, protected from light. Keep out of the reach of children. Do
not touch dropper tip to any surface as this may contaminate Iventi. Do not use
after one month of first opening.
Frequently Bought Products
Emjard Tablet, Empagliflozin 10 mg
Timotor Tablet, Trimebutine Maleate 100 mg
Itra Capsule, Itraconazole 100 mg
Comprid XR Tablet (Extended Release) , Gliclazide 60 mg
Gutfix Capsule, Lubiprostone 8 mcg
Mevin Tablet, Mebeverine Hydrochloride 135 mg
Product Queries (0)
Login Or Registerto submit your questions to seller
Other Questions
No none asked to seller yet
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00