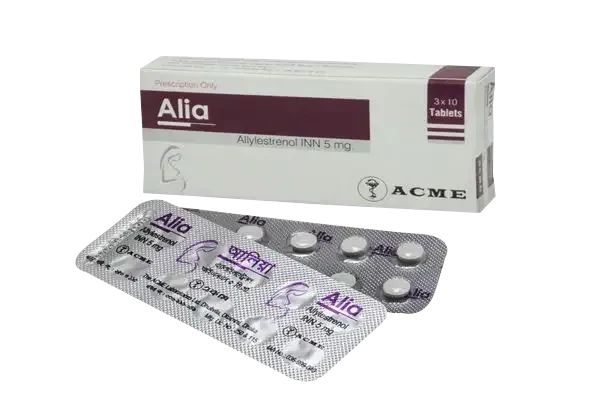

Alia Tablet, Allylestrenol 5 mg
Inhouse product
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00
Reviews & Ratings
Indications
Alia is indicated in:
- Intra Uterine Growth
Retardation (IUGR)
- Threatened abortion
- Habitual abortion
- Threatened premature delivery
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Description
Alia is an oral
Gestagen. In castrated animals, it has a potent pregnancy-maintaining action
without producing hormonal side-effects, in particular no masculinisation of
female foetuses. Clinical studies have indicated that Alia stimulates the
endocrine function of the trophoblast. In vitro, Alia stimulates the placenta
to produce more progesterone. In patients with pregnancy at risk, abnormally
low levels of placental hormones (HCG, HPL, oestrogens and progesterone) and
some specific enzymes (oxytocinase, CAP) were found to be increased to normal during
treatment. In the majority of patients with pregnancy at risk who were treated
with Alia the threat of abortion was removed and pregnancy brought to term
without complications. Alia is very well tolerated. Side-effects of any
importance have not been observed.
Pharmacology
Allylestrenol has been
found to have a relatively weak progestational effect on the human endometrium.
To obtain a full secretory endometrium in oestrogen-primed castrated women or
to postpone menstruation (with an oestrogen added) in normal ovulating women,
doses of allylestrenol were required which were higher than those recommended
for the treatment/prevention of abortion.
In vitro studies have shown that allylestrenol stimulates the synthesis of
progesterone in the human placenta. It also brought about a significant (p/.
0,01) increase in the production of some specific placental enzymes (cystine
aminopeptidase and heat-stable alkaline phosphatase).
Histological and histochemical changes indicating an increased activity have been
found in the placenta, particularly in the syncytiotrophoblast of women with a
normal and threatened pregnancy, treated with allylestrenol. The stimulatory
effect of allylestrenol on placental function was also suggested by the
increased level of placental hormones (pregnanediol, oestriol, HCG and HPL) and
enzymes (oxytocinase, CAP) in the maternal urine and plasma, which followed the
administration of the drug e.g. in the early weeks as well as in the last
trimester of pregnancy.
A study in full-term pregnant women revealed that allylestrenol in high doses
(up to 100 mg daily) did suppress the intensity of spontaneous uterine
contractions, but had no effect on the sensitivity of the uterine muscle to
oxytocin, and no adverse effect on the progress of normal delivery.
Studies in non-pregnant women with and without endocrinological disorders have
shown that allylestrenol has no oestrogenic or androgenic properties and no
adverse effects on the adrenal function.
No abnormal liver function tests or water and salt retention were observed in
healthy female volunteers (non pregnant) who were given allylestrenol.
Dosage &
Administration
Intra Uterine Growth
Retardation: 1 tablet three times
a day at least two months. Dose to be reduced if symptoms improve.
Threatened abortion: 1 tablet three times daily until symptoms
disappear.
Habitual abortion: 1-2 tablets daily as soon as pregnancy is
diagnosed. The administration should be continued for at least one month after
the end of the critical period.
Threatened premature
delivery: Dosage must be
determined individually. High dosages (up to 40 mg daily) have been used.
In case of a missed dose, it should be taken as soon as the patient remembers
& she should continue the regular dosing schedule. A double dose is not
recommended.
* রেজিস্টার্ড চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Contraindications
- Breast Cancer or history of
problem with the breasts like- Nodules,an abnormal Mammogram, or
Fibrocystic Diseases.
- Severe liver disease such as
Cholestatic Jaundice or Hepatitis, Hepatic Cell Tumours, Rotor Syndrome
and Dubin Jhonson Syndrome.
- Undiagnosed vaginal bleeding
- Toxaemia of Pregnancy
- Crisis Seizures, Migraines
Side Effects
Treatment with Alia
(especially a long term treatment) is known to cause some gastrointestinal
complaints such as vomiting, nausea, and sometimes epigastric discomfort.
Pregnancy &
Lactation
Allylestrenol is
specifically designed to be taken during pregnancy. It should be discontinued
after delivery as it may affect a nursing infant to a small but noticeable
degree.
Precautions &
Warnings
Patients with the
following conditions should be cautious: Heart disease, congestive heart
failure, sick sinus syndrome, coronary artery disease, seizures, epilepsy,
renal dysfunction, migraine headaches, or breathing diseases including asthma,
emphysema, chronic bronchitis, or COPD, breast-feeding.
Use in Special
Populations
It should not be used
for children younger than 16 years old.
Overdose Effects
Symptoms of overdose
may include unusual drowsiness; rapid pulse; fainting; unusual muscle movement
or rigidity of the face, neck, or limbs; seizures; and loss of consciousness.
Therapeutic Class
Female Sex hormones
Storage Conditions
Keep below 30°C
temperature, away from light & moisture. Keep out of the reach of children.
Frequently Bought Products
Leviceta Table, Levetiracetam 500 mg
Cipro-A IV Infusion 100 ml bottle, Ciprofloxacin 200 mg/100 ml
Dermupin Ointment 15 gm tube, Mupirocin 2% w/w
Fast Pediatric Drops 15 ml bottle, Paracetamol 80 mg/ml
Ranola Tablet (Extended Release), Ranolazine 500 mg
Egolix Tablet, Elagolix Sodium 150 mg
Product Queries (0)
Login Or Registerto submit your questions to seller
Other Questions
No none asked to seller yet
-
৳11.40
৳12.00 -
৳42.75
৳45.00 -
৳16.63
৳17.50 -
৳2.14
৳2.25 -
৳9.50
৳10.00